SodaStream and Hadassah Medical Center collaborate on new respiratory device
By PLANT STAFF
Industry Manufacturing COVID-19 Hadassah Medical Center manufacturing Respirator SodaStream StreamO2The clinical trial of StreamO2 has now started in the Jerusalem Hospital after receiving approval by Israeli Ministry of Health
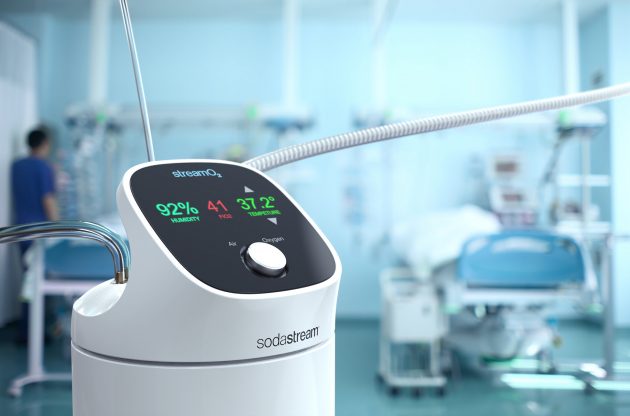
SodaStream StreamO2. PHOTO: SodaStream
MISSISSAUGA — A solution inspired by SodaStream technology in collaboration with the Hadassah Ein Kerem Hospital will enable respiratory assistance for COVID-19 patients in mild and moderate conditions.
At the core of this collaboration stands the development of a medical solution for nasal breathing therapy. The device has been given approval by the Israeli Ministry of Health and a clinical trial in the general intensive care unit has already begun.
According to a company statement, the innovation is designed to treat COVID-19 patients in mild to moderate conditions who are in respiratory distress but still breathing spontaneously. The device provides patients with the right levels of oxygen required in their situation. Studies have shown that this type of oxygen device can prevent aggravation and intubation (invasive respiratory assistance) in some patients, and may reduce the risks and complications associated with it.
In addition, the device may accommodate the shortage of respiratory machines, and make them available for patients in respiratory distress for whom the help of an invasive respiratory aid machine is the only available treatment option.
The device developed by SodaStream and Hadassah allows for the administration of an oxygen-air mixture through the nose at high flow rates, high humidity, and body temperature. The development is based on a thermodynamic system that controls the temperature of the air-oxygen mixture and flows the mixture into the patient’s nose in combination with high humidity of over 80%. The parameters of humidity, temperature, and percentage of oxygen are monitored and provide a visual and audible indication to the medical staff in case of abnormality.
While a small number of similar products currently exist in the market and meet the above requirement, they are limited for individual treatment, and not for mass treatment situations such as a pandemic.
With the outbreak of COVID-19, Akiva Nachshon, a senior physician in the intensive care unit at Hadassah Ein Kerem Hospital, contacted Avi Cohen, head of the research and development division of SodaStream, and they set up an engineering team comprised of SodaStream development engineers. The engineering team — led by Cohen with the medical guidance of Nachshon — designed and built the machine while adhering to the strictest technology and regulatory requirements.
Due to the urgent necessity for the product, the team received an accelerated approval from the Clinical Trials Unit of the Israeli Ministry of Health – and just initiated the clinical trial comprised of 40 patients.
“We realized that there was an acute shortage of non-invasive respiratory aids to treat COVID-19 patients, and after recognizing that there was a desire from the industry to help in dealing with this crisis, we tried to think outside the box”, explained Nachshon in a prepared statement. “We learned based on a friend’s advice that the solution is right here in the Israeli industry, under our nose, in every kitchen. One conversation with SodaStream Head of R&D was enough to get things going. On the same day, we already sat on a sketch, and from that point the project progressed incredibly fast.”